Certified clinical trials: digital evidence for trial monitoring and compliance
Certify clinical trial documentation at the source with digital signature and timestamp for GCP and ALCOA+ compliant data.
Clinical trials require rigorous, traceable documentation at every stage. EU Regulation 536/2014 and ICH E6(R3) guidelines set demanding standards for the collection, preservation and verifiability of clinical data, in line with ALCOA+ principles.
Site activity photographs, source documents, adverse event records and investigational medicinal product accountability checks often lack guarantees of authenticity and certain time-stamping. TrueScreen certifies every digital evidence at the moment of collection with a digital signature and timestamp, ensuring integrity, traceability and regulatory compliance from the source.
TrueScreen enables sponsors, CROs and trial sites to certify trial documentation directly at the point of collection. Every photo, video, source document and monitoring report is digitally signed and timestamped, ensuring compliance with the ALCOA+ principles required by GCP.
Industry
Pharmaceutical, clinical research, CRO
Business function
Clinical operations, quality assurance, regulatory affairs
Key process
Clinical trial documentation and monitoring
Certified content
Photos, videos, source documents, monitoring reports, recordings, checklists
Output
Certified documentation and reporting with legal validity
Adoption mode
App / Web / API / SDK
Needs

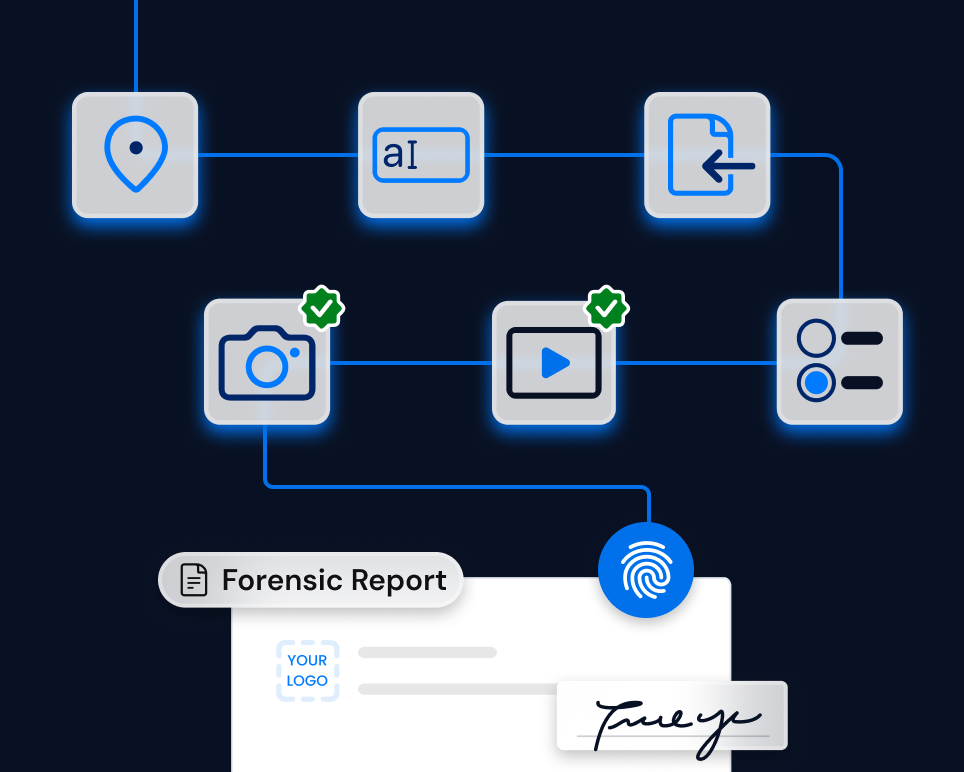
Solution
TrueScreen enables sponsors, CROs and clinical trial sites to certify photos, videos, source documents and monitoring reports directly during trial operations.
Every piece of content is certified with immutable metadata: timestamp, GPS coordinates and operator identity. Source data verification reports and monitoring visit records are digitally signed, creating a certified dossier for each trial, site or regulatory submission.
In the event of a regulatory inspection or audit, the certified dossier provides evidence with guaranteed provenance and integrity, reducing the risk of findings and simplifying source data verification across clinical sites.
What gets certified
Site documentation
Certified photos and videos of trial site activities, investigational pharmacy, drug storage conditions and visit areas
Source documents
Certified acquisition of CRFs, informed consents, laboratory reports, patient diaries and any original trial document
Monitoring reports
Certified documentation of monitoring visits, on-site verifications, protocol deviations and corrective actions
Drug accountability
Certified records of receipt, dispensing, return and destruction of the investigational product with complete traceability
Partners
Pharmaceutical companies, CROs (Contract Research Organizations), trial sites, ethics committees, regulatory authorities, regulatory consulting firms.
Integrations
eTMF systems (Electronic Trial Master File), EDC platforms (Electronic Data Capture), CTMS systems (Clinical Trial Management System), pharmacovigilance software, quality management systems.
FAQ: certified clinical trials and trial documentation
1) How does clinical trial documentation certification work?
2) Is TrueScreen certification compliant with ICH E6(R3) GCP?
3) What trial documents can I certify?
4) How does it support source data verification?
5) How does it integrate with eTMF and EDC systems?
6) How does it reduce the risk of inspection findings?
Request a free demo
Talk to our experts and discover TrueScreen for certified clinical trial documentation.
