Certified medical devices: digital evidence for traceability and surveillance
Certification of inspections, maintenance and vigilance reports on medical devices with legally valid digital evidence.
Manufacturers, distributors and healthcare operators managing medical devices must ensure traceability, technical documentation and post-market surveillance throughout the product lifecycle. The European MDR Regulation (EU 2017/745) requires unique device identification (UDI), documentation retention for at least 10 years, and incident reporting.
Photos, videos and documents collected during inspections and maintenance often lack certain dates and integrity guarantees. TrueScreen certifies every documentation phase with immutable metadata, timestamps, cryptographic hash and digital signature, producing legally valid evidence compliant with the eIDAS Regulation.
Industry
Medical devices, healthcare, biomedical, pharmaceutical
Business function
Quality and regulatory affairs, Clinical engineering, Biomedical maintenance, Vigilance, Legal department
Key process
Inspection and verification certification, maintenance documentation, vigilance reports, lifecycle traceability, post-market surveillance
Certified content
Photos, videos, operational checklists, inspection reports, maintenance reports, incident reports, technical documentation
Output
Certified documentation and reporting with legal validity
Adoption mode
App / Web / API / SDK
Needs

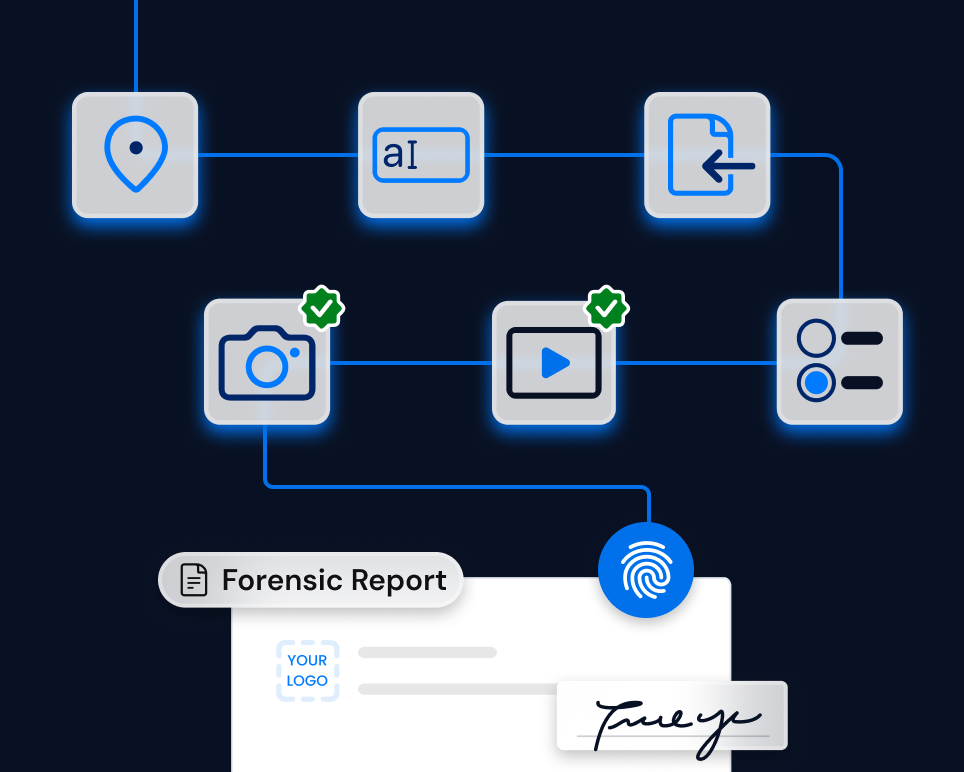
Solution
TrueScreen integrates into medical device management processes, enabling clinical engineers, maintenance technicians and quality managers to certify every documentation phase directly in the field.
Photos of device conditions, verification videos, completed checklists and inspection reports automatically receive immutable metadata: timestamps, geolocation, cryptographic hash and digital signature compliant with the eIDAS Regulation.
The certified documentation is opposable during competent authority inspections, in vigilance reports and in legal proceedings, ensuring compliance with MDR Regulation obligations throughout the entire device lifecycle.
What gets certified
Medical device condition
Photos and videos of operating conditions, components and UDI labelling
Maintenance interventions
Documentation of operations performed with certified checklists and chronological register
Periodic inspections and verifications
Inspection reports with detection of conditions and measurements performed
Vigilance reports
Photographic and video documentation of anomalies detected with certain date
Partners
Medical device manufacturers, authorised distributors, public and private healthcare facilities, clinical engineering services, biomedical maintenance companies, notified bodies, certification bodies and law firms specialised in healthcare law can integrate TrueScreen into their operational workflows.
Integrations
TrueScreen integrates with quality management systems, biomedical CMMS, post-market surveillance platforms, EUDAMED databases and document archives. Certified reports are exportable in PDF and JSON format, compatible with major archiving systems and reporting platforms for competent authorities.
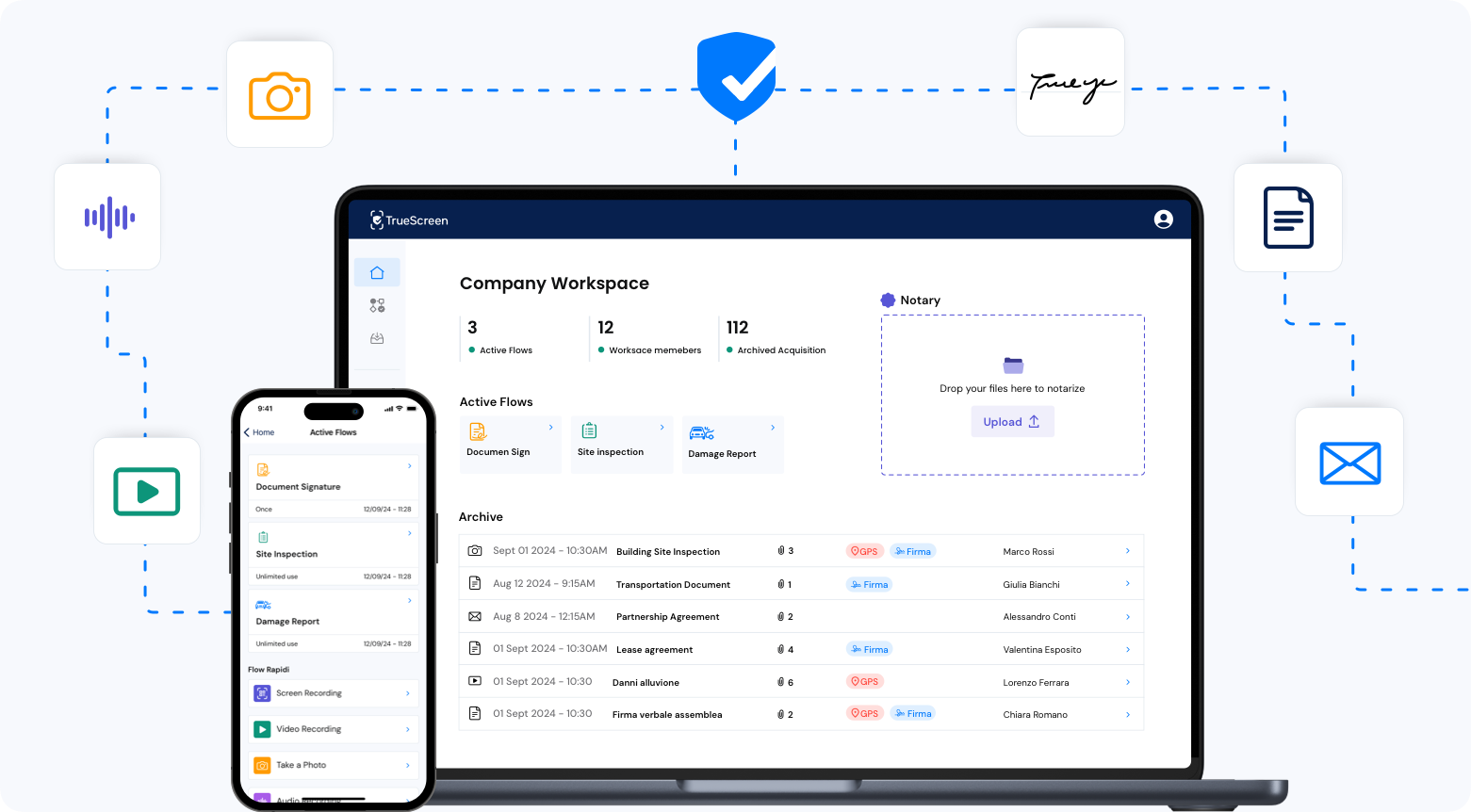
FAQ: certified medical devices and traceability
1) How does TrueScreen certify medical device documentation?
2) Is TrueScreen certification compliant with the MDR Regulation (EU 2017/745)?
3) What types of medical device documents can be certified?
4) How does UDI traceability work with TrueScreen certification?
5) How are vigilance reports managed with certified evidence?
6) Can certified reports be integrated into quality management systems?
Request a free demo
Talk to our experts and discover TrueScreen for certified medical device documentation.
