Pharmaceutical and Medical Device Verification: Certified Digital Evidence
Forensic certification of inspections, audits and verifications to ensure document integrity, supply chain traceability and regulatory compliance.
The pharmaceutical and medical device industry operates under increasing regulatory pressure. Every inspection, every compliance verification, every supply chain audit generates digital evidence: batch photographs, inspection videos, serialization documents, non-conformity reports. This evidence must be authentic, unaltered and traceable to withstand scrutiny from regulatory authorities. With pharmaceutical counterfeiting generating billions in illicit revenue and European regulations (MDR, EU Reg. 2016/161) requiring increasingly rigorous documentation, relying on photographs and reports without authenticity guarantees exposes organizations to legal risks, sanctions and disputes.
TrueScreen certifies every piece of evidence at the point of collection, with digital signature, qualified timestamp and verified metadata, transforming verification activities into documented processes with full legal validity.
Industry
Pharmaceutical and medical devices
Business Function
Protezione civile, Operations, Sinistri e liquidazione, Risk management, Compliance, Legale
Key Process
Valutazione danni, gestione interventi e ripristino, gestione sinistri e rimborsi, audit post-evento
Certified Content
Foto, video, audio, screenshot, navigazione web, verbali di sopralluogo
Output
Certified documentation and reporting with legal validity
Adoption Mode
App / Web / API / SDK
Needs

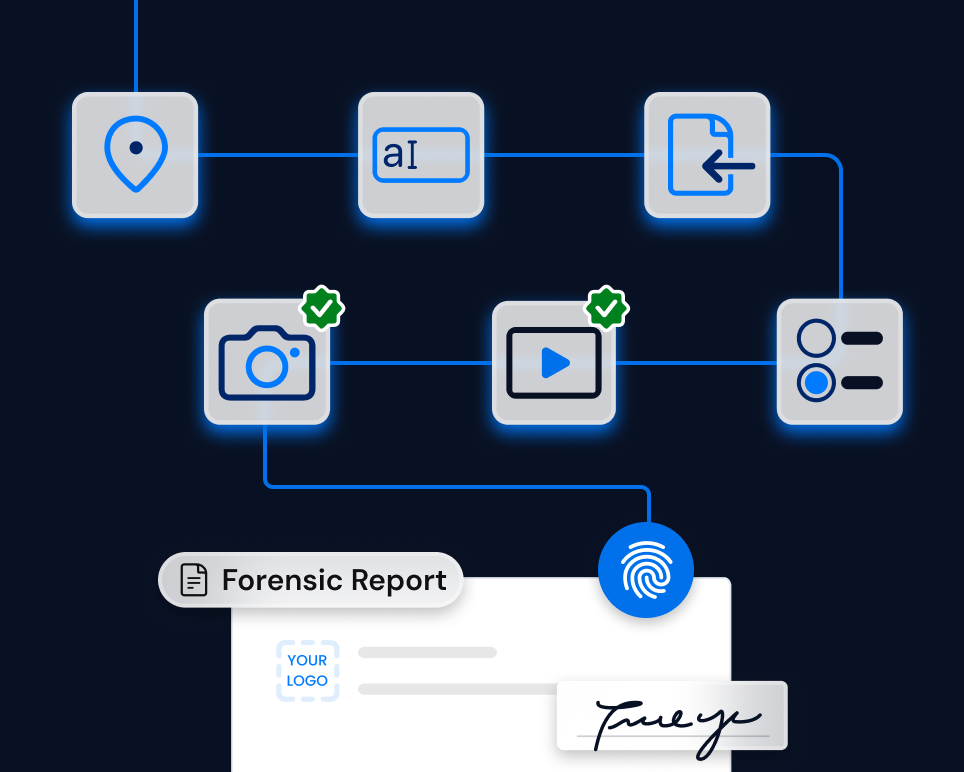
Solution
TrueScreen enables inspectors, Quality Assurance managers and field operators to certify every piece of digital evidence at the point of collection. Every batch photograph, every inspection video, every traceability system screenshot is captured with verified metadata (GPS position, date and time, device), digitally signed and equipped with a qualified timestamp compliant with eIDAS.
The result is verification documentation with a complete and unbroken chain of custody, ready for regulatory audits, legal proceedings and compliance checks.
What gets certified
Batches and Packaging
Photographs of batches, packaging, labels and Data Matrix codes for serialization verification with certified position metadata and timestamps
Video Inspections
Video recordings of inspections in manufacturing facilities, warehouses and distribution centers with intact chain of custody from recording
Traceability Systems
Screenshots of traceability systems and digital supply chain records, environmental condition recordings during storage and transportation
Reports and Audits
Non-conformity documents, pharmacovigilance reports and audit reports digitally signed with immutable metadata and legal validity
Partners
Pharmaceutical companies, medical device manufacturers, distributors and wholesalers, notified bodies, regulatory consulting firms, testing and quality control laboratories.
Integrations
Quality Management Systems (QMS) compliant with ISO 13485, serialization and traceability platforms (EU FMD, US DSCSA), pharmaceutical ERP systems, pharmacovigilance platforms, document archives compliant with 21 CFR Part 11.
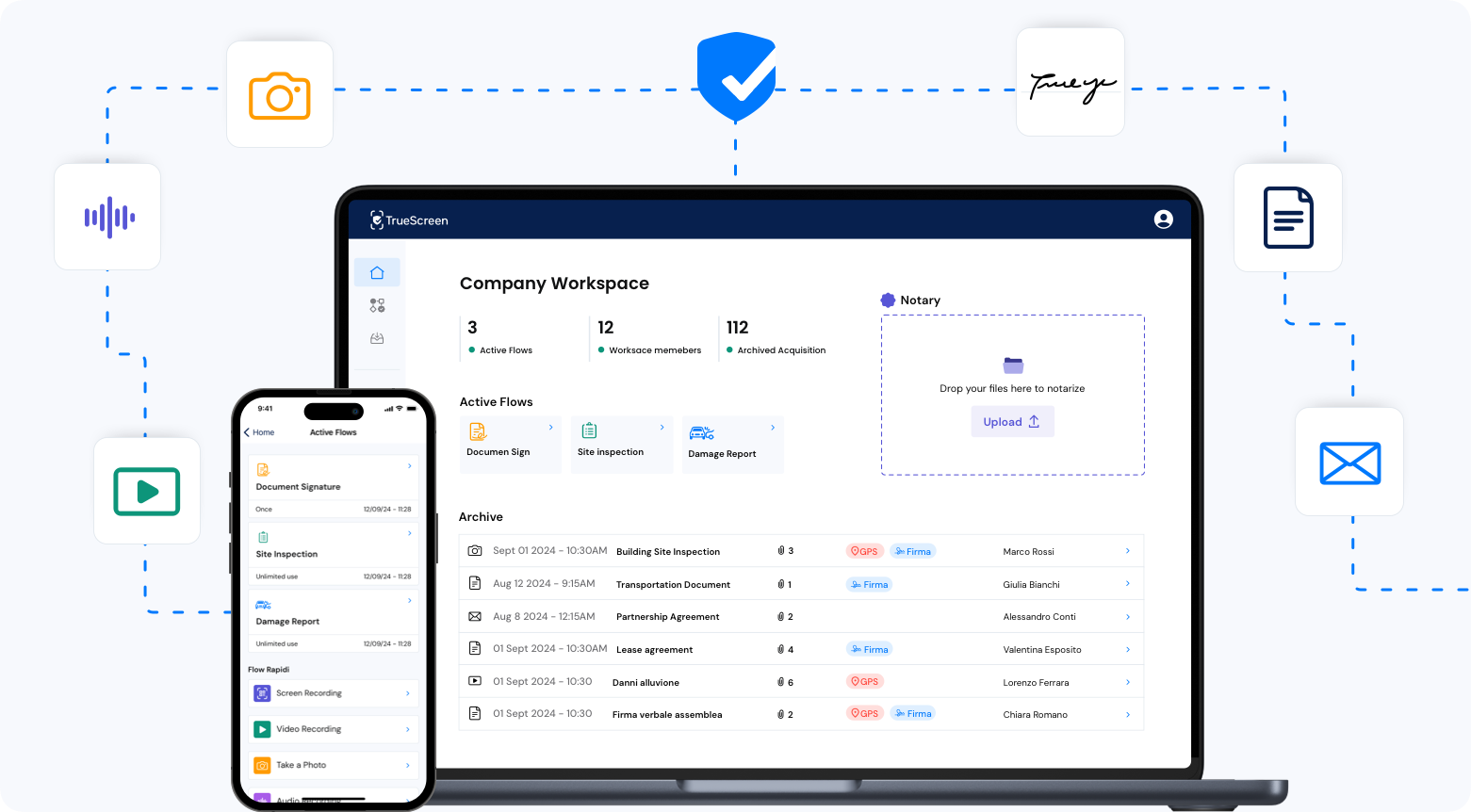
FAQ: Certified Pharmaceutical and Medical Device Verification
1) What types of digital evidence can be certified during a pharmaceutical inspection?
2) How does TrueScreen ensure compliance with MDR 2017/745 requirements?
3) How does certification protect the chain of custody for inspection evidence?
4) How much time is saved in producing audit documentation?
5) Can certified documentation be used in legal proceedings?
6) How does TrueScreen integrate with existing Quality Management systems?
Request a demo gratuita
Talk to our experts and discover TrueScreen for certified pharmaceutical and medical device verification.
